Web Menu
Product Search
Exit Menu
We provide quality products and services to customers from all over the world.
Sports Adhesive Tapes: Production and Challenges
Sports adhesive tapes are widely used in athletic and medical settings to provide support, prevent injuries, and stabilize joints. These tapes combine material science and manufacturing techniques to produce products that adhere to skin and equipment while remaining flexible.

Processes Involved in the Formation of Sports Adhesive Tapes
The manufacturing of sports adhesive tapes involves several sequential processes, from material preparation to final quality control. Some key steps include:
Material Selection and Preparation
Base Material: The backing of the tape is usually made from cloth, synthetic fibers, or elastic materials, depending on flexibility and strength requirements.
Adhesive Composition: The adhesive is typically pressure-sensitive, formulated from natural or synthetic rubber, acrylic compounds, or other polymers to provide consistent adhesion without causing skin irritation.
Additives: Plasticizers, tackifiers, and stabilizers are incorporated to improve elasticity, durability, and shelf life.
Coating and Lamination
Adhesive Application: The adhesive is applied to the backing material using techniques such as roller coating or spraying. Uniform application ensures consistent stickiness and prevents gaps.
Lamination and Drying: After adhesive application, the tape may undergo lamination or heating to bond layers securely and remove excess moisture.
Cutting and Slitting: Large rolls of tape are cut into standardized widths and lengths suitable for retail or professional use.
Quality Control and Packaging
Adhesion Testing: Tapes are tested for stickiness, flexibility, and peel strength to ensure performance meets specifications.
Packaging: Finished tapes are rolled, labeled, and packaged under controlled conditions to prevent contamination or damage.
These processes ensure that sports adhesive tapes are durable, flexible, and suitable for repeated use in athletic or medical contexts.
Problems with Sports Adhesive Tapes
Despite their widespread use, sports adhesive tapes have limitations and potential issues that affect performance and user experience.
Skin Irritation and Allergic Reactions
Irritation: Prolonged application can cause redness, itching, or discomfort, particularly in sensitive skin areas.
Allergic Reactions: Some users may react to adhesives containing latex or certain chemical additives, resulting in rashes or blisters.
Adhesion and Durability Issues
Loss of Stickiness: Sweat, water, or prolonged movement can reduce adhesion, causing the tape to peel or slide.
Elasticity Loss: Tapes may stretch beyond their capacity, reducing support or failing to maintain tension over time.
Surface Contamination: Dust, dirt, or oils on the skin can interfere with proper adhesion, making the tape less effective.
Application and Removal Challenges
Incorrect Application: Tapes applied too tightly may restrict blood flow, while loose application may fail to provide sufficient support.
Painful Removal: Removing adhesive tape can be uncomfortable and may damage the skin or hair, particularly after long-term use.
Limited Reusability: Many sports adhesive tapes are designed for single-use, generating waste and requiring frequent replacement.
Environmental and Storage Considerations
Temperature Sensitivity: Heat or cold can affect adhesive performance.
Storage Conditions: Poor storage may cause the tape to dry out or lose stickiness before use.
These factors highlight that while sports adhesive tapes are effective tools for injury prevention and support, careful attention is required during selection, application, and storage to minimize problems.
Sports adhesive tapes are produced through a series of controlled processes, including material preparation, adhesive coating, lamination, and quality testing. These processes ensure the tapes provide support, flexibility, and reliable adhesion for athletes and medical users. However, challenges such as skin irritation, reduced adhesion, application errors, and environmental sensitivity can limit effectiveness. Understanding both the production process and potential problems helps users and manufacturers optimize the use of sports adhesive tapes while minimizing discomfort and performance issues.
Keep In Touch
No.455 New Town Development Road,Longgang City, Wenzhou City
Copyright © Wenzhou Anqi Medical Supplies Co., Ltd. All Rights Reserved.
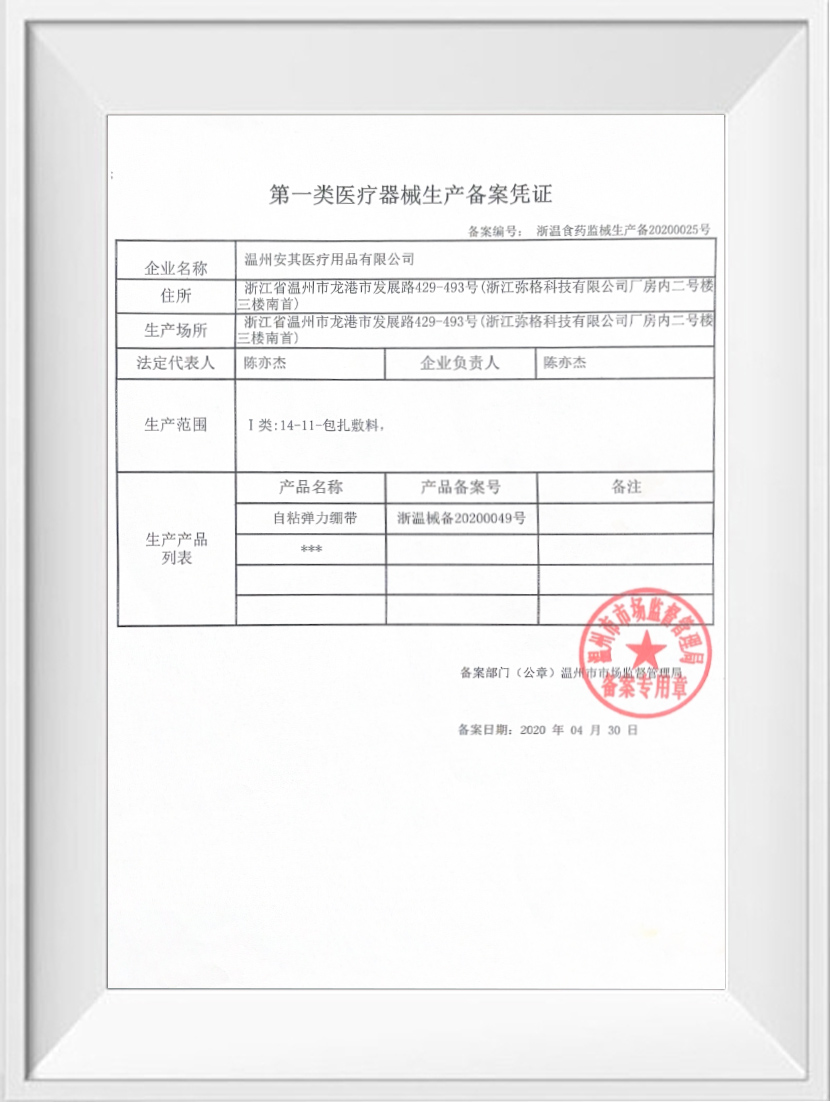
Approval No: Zhejiang Wenzhou Food and Drug Administration Machinery Production Filing No. 20200025
The information provided on this website is intended for use only in countries and jurisdictions outside of the People's Republic of China.

 English
English русский
русский 中文简体
中文简体 Español
Español Deutsch
Deutsch عربى
عربى